News
CM5 and Midterm Workshop – Yes, it really is that difficult.
Wed. 17. Oct 2018 12:00
ImpleMentAll’s 5th Consortium Meeting (CM5) took place in Odense, Denmark, on 8-9 October 2018 flanked by a couple of satellite meetings and the project’s Midterm Workshop. While closely related and interdependent, each of these events had their own distinct purpose and audience, as reported below.
Scientific Steering Committee Meeting
The role of the IMA SSC is to support the Scientific Coordinator, Christiaan Vis, and supervise the research carried out in the project, ensuring the high level of scientific quality nedded to succeed with the ambitious goals of the project. The SSC is led by prof. dr. Heleen Riper from the Vrije Universiteit Amsterdam and composed of Prof. dr. Carl May, Prof. dr. Helen Christensen, Prof. dr. Marion Leboyer, Prof. dr. Ulrich Hegerl, Prof. dr. Jan Smit, Associate Prof. Kristian Kidholm, dr. David Ebert, and the Project Coordinator mr. Claus Duedal Pedersen.
The SSC meeting was held in the morning before the start of the CM and focused on three topics related to 1) the importance of trial integrity, 2) providing guidance of the trial sites in applying the ItFits toolkit, and 3) the publication procedure, acknowledgment, and authorship. These topics represent some of the most critical issues faced by IMA as we have started collecting data and will see the first two sites crossing over to ItFits application before the end of the year. The SSC members were very helpful in their feedback and advice, and helped the Scientific Coordinator and team overcome some of the challenges that come from running a trial as complex as ours. Read more about the theoretical background of the project and its study protocol on the blog.
5th Consortium Meeting
The CM5 was the first Consortium Meeting after Reporting Period 1, where the project delivered its first progress report to the European Commission detailing its progress over the project’s initial 18 months. It was also the first CM after the beginning of data collection on 1 June 2018, which has given the consortium, and especially the Coordination Team and Trial Management, a good indication of both the status and feasibility of the coming trial and ItFits application. From 1 December 2018, the ItFits toolkit will be trialled in the first two sites to cross over from Implementation-as-Usual to implementation guided by the toolkit.
The meeting focused on recruitment of staff, data collection, data quality, communication and dissemination, the ItFits-toolkit, the first cross-over, the process evaluation, and stakeholder involvement through the External Advisory Board. As the first cross-over is nearing, there is a sense of anticipation in the consortium and the CM contributed to clearing up many of the last questions and doubts remaining before the trial begins in earnest.
The next CM will be held in Turin, Italy on 7-8 May 2019.
External Advisory Board Meeting
The IMA EAB provides expert advice from many different angles (policy, research, dissemination, exploitation, etc.) to ensure that the project is developing in the right direction according to its ambitions. The EAB is managed by the WP6 team from RSD and composed of Bianca Albers (EIC), Bruce Whitear (tCI), Clayton Hamilton (WHO), Chris Wright (NHS 24), David C. Mohr (Northwestern University Feinberg School of Medicine), Dean L. Fixsen (University of North Carolina at Chapel Hill), Elizabeth Murray (University College London), Genc Burazeri (Tirana Medical University), Hobbe Jan Hiemstra (E-Mence), John Crawford (Digital Health Consultant), Levente Kriston (University Medical Center Hamburg-Eppendorf), Mark Bloemendaal (ImplementationIQ), Markus Moessner (Center for Psychotherapy Research), Nick Titov (Macquarie University), Richardo Gusmao (EUTIMIA), Simone Gynnemo (patient, Health care pedagogue and Psychogenesis therapist), and Vicente Traver Salcedo (SABIEN, ITACA Institute).
The meeting focused on questions raised by IMA WP leaders for the EAB, such as “Qualitative process evaluation of Itfits: are we asking (all of) the right questions?” and “To what extent can we adapt to changes that happen in real-world conditions in trial sites?”. The meeting was the first occasion for the EAB to meet in person, although some members joined virtually. The discussions were very concrete and the WP leaders gained helpful advice and inspiration for their specific tasks and challenges.
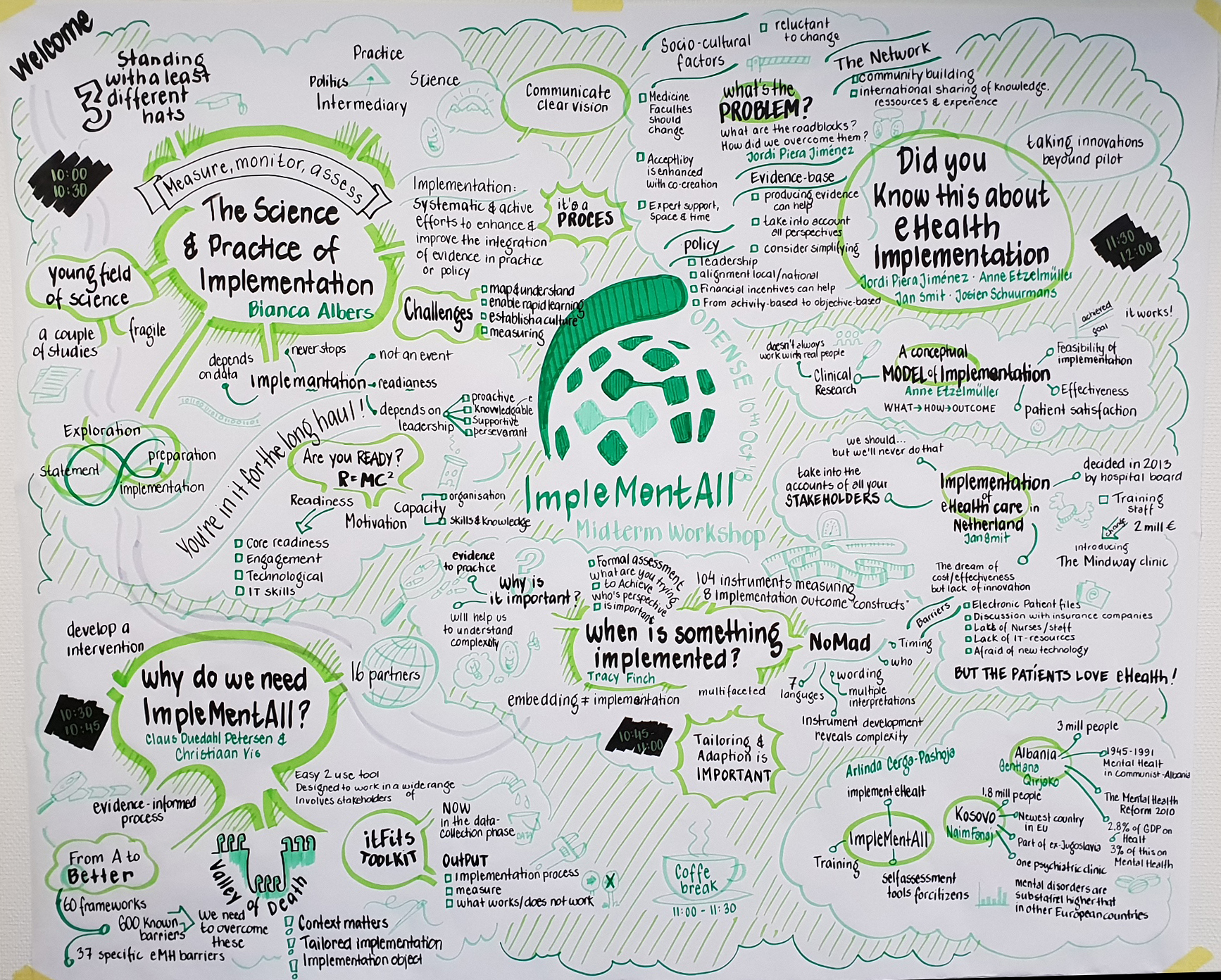
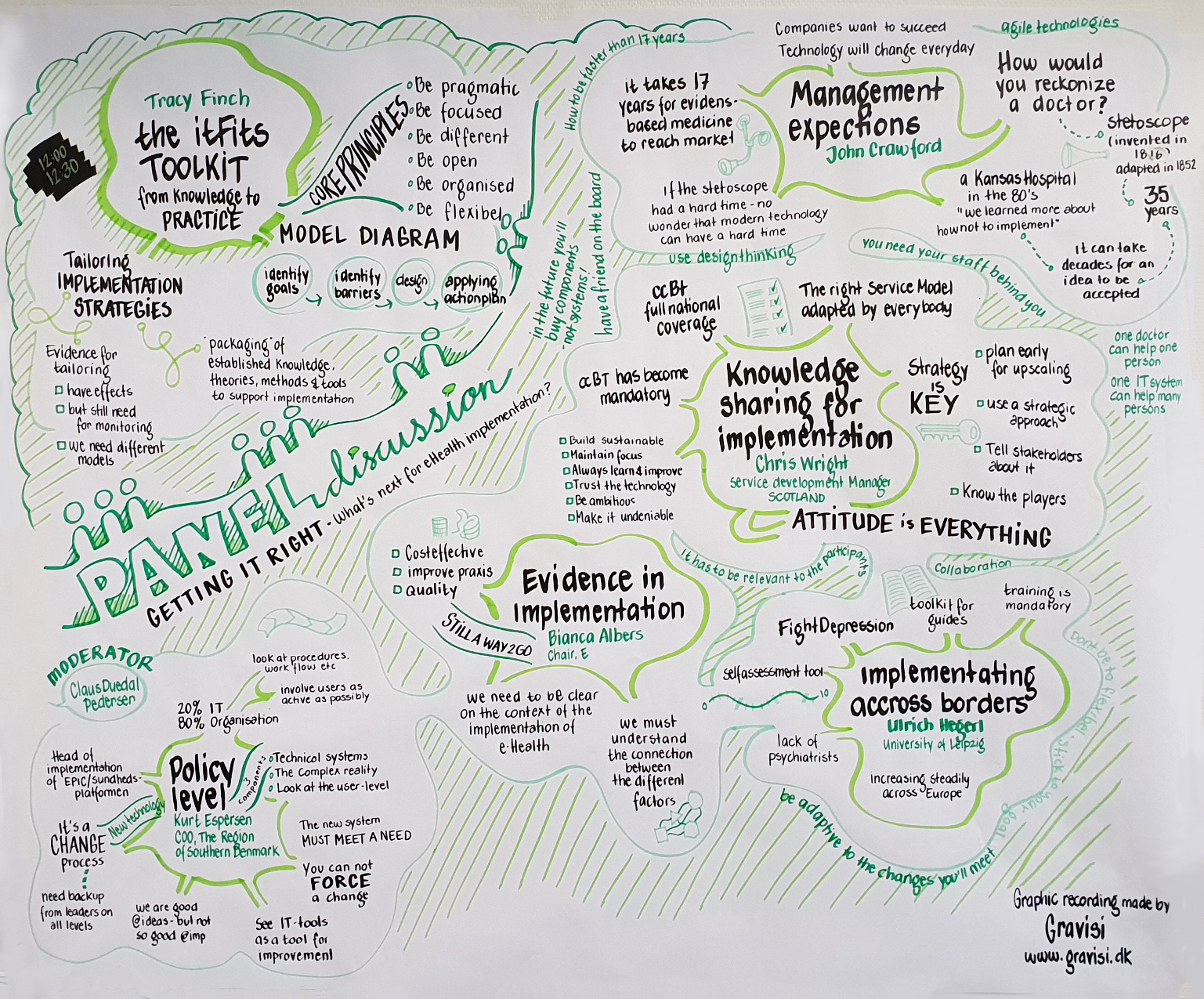
Midterm Workshop
The purpose of the Midterm Workshop was to showcase the work of the project and to debate the most critical issues related to implementation science and practice. The workshop was held as part of the WHINN conference in Odense and at a point in time where the project is moving from trial preparation to trial operation. To support its purpose, the workshop was built on three main questions: “Implementation science – changing the world one barrier at a time?”, “Did you know this about eHealth implementation?”, and “Getting it right – what’s next for eHealth implementation?”.
By including the workshop in the programme of an international conference, the project was able to draw in a much wider range of stakeholders than if it had been a stand-alone event. This enhanced the discussions as experiences and knowledge were shared between the consortium, the speakers, and the rest of the participants. One of the main points that pervaded the discussions was the statement “Is implementation really that difficult? Yes, it is.”. Still, this doesn’t mean that there is no hope. ItFits, which is a digitally accessible toolkit for tailoring implementation activities to key barriers, was presented as IMA’s answer to the problem. One of its main components, tailoring, is a systematic process of identification of local determinants, selecting, and designing implementation strategies, subsequently applying and measuring their effectiveness. This requires being able to determine when/if something is ‘implemented’, which relies on a balance of science, pragmatism, and genuine stakeholder co-production. Finally it was put forward that implementation is one thing, but sustaining an innovative service or treatment is the tricky part and an effort which never really ends.
At the workshop, it was revealed that, after many requests, the translations of ORIC and NoMAD will be published on the website as one of the first outcomes of IMA. These are expected to be online within the coming weeks.
The speakers included both IMA consortium members and external stakeholders (click for slides): Bianca Albers (EIC), Christiaan Vis (VUA), Claus Duedal Pedersen (RSD), Tracy Finch (UNN) session 1 & session 2, Jordi Piera Jiménez (BSA), Anne Etzelmüller (Get.ON), Jan Smit (GGZ inGeest), Arlinda Cerga-Pashoja (LSHTM), Naim Fanaj (MHC, Prizren), Gentiana Qirjako (IPH, Tirana), Kurt Espersen (RSD), Chris Wright (NHS 24), Ulrich Hegerl (EAAD), and John Crawford (Digital Health consultant). See the full programme here.
After these three days of intense discussions and dissemination of the project’s work, it was clear that the thoughts behind the project and the work done so far are a good match for what is needed in eHealth implementation – and that no one has solved the problem while we have been designing and setting up our trial. The ItFits-toolkit, with the knowledge built into it, will be useful for implementation efforts in any kind of health organisation across Europe and globally, offering concrete, tailored guidance in the rigth direction. Last but not least, it once again became evident that the mere catalyst effect of partners coming together in a project of this kind can move mountains.